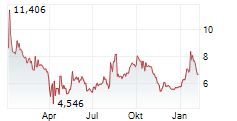
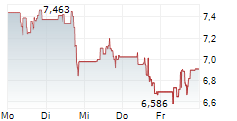
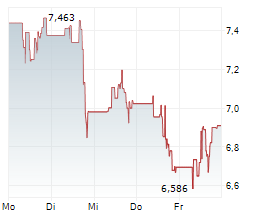
WASHINGTON (dpa-AFX) - Novavax, Inc. (NVAX) announced that the Medicines and Healthcare products Regulatory Agency or MHRA in the United Kingdom has granted full marketing authorization for its prototype COVID-19 vaccine Nuvaxovid or NVX-CoV2373 for individuals aged 12 and older for active immunization to help prevent COVID-19.
Separately, Novavax, Inc. announced that Singapore's Health Sciences Authority has granted full approval for Novavax's prototype COVID-19 vaccine Nuvaxovid or NVX-CoV2373 for active immunization to prevent COVID-19 in individuals aged 12 and older.
'Today's full approval of our prototype vaccine will enable us to file for approval of our updated protein-based non-mRNA COVID-19 vaccine in the coming weeks,' said John Jacobs, CEO, Novavax.
For More Such Health News, visit rttnews.com.
Copyright(c) 2023 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX