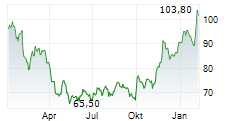
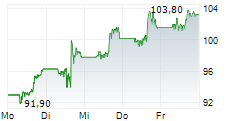
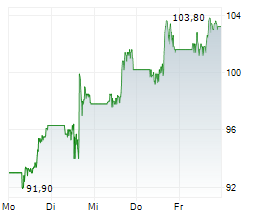
KENILWORTH (NJ) (dpa-AFX) - Merck (MRK) announced the FDA has accepted and granted priority review for a supplemental new drug application seeking approval for WELIREG for the treatment of adult patients with advanced renal cell carcinoma following immune checkpoint and anti-angiogenic therapies. The FDA has set a PDUFA, or target action, date of January 17, 2024.
WELIREG was the first HIF-2? inhibitor therapy approved in the U.S. It is currently approved for the treatment of adult patients with von Hippel-Lindau disease who require therapy for associated RCC, central nervous system hemangioblastomas, or pancreatic neuroendocrine tumors, not requiring immediate surgery.
For More Such Health News, visit rttnews.com.
Copyright(c) 2023 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX